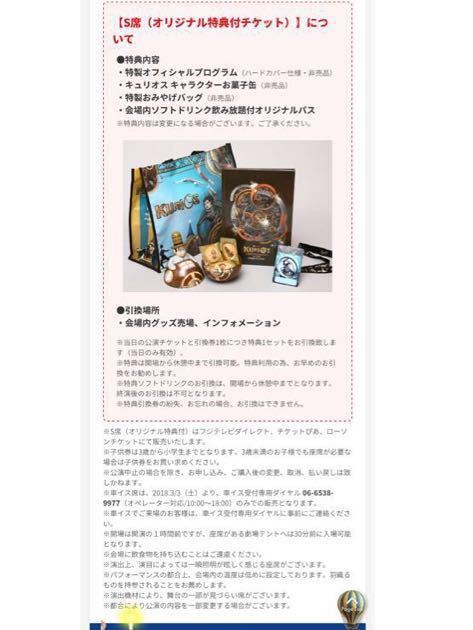
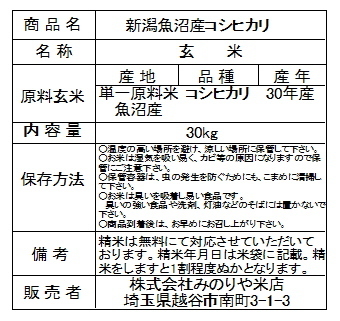
★ 大容量2.3kg ★ Optimum Nutrition社 100%ホエイ ゴールドスタンダード プロテイン ★
(460件)
Pontaパス特典
サンキュー配送
4470円(税込)
153ポイント(1%)
Pontaパス会員ならさらに+1%ポイント還元!
送料
(
)
22
配送情報
お届け予定日:05月29日までにお届け
※一部地域・離島につきましては、表示のお届け予定日期間内にお届けできない場合があります。
ロットナンバー
P2143983937
お買い物の前にチェック!

Pontaパス会員なら
ポイント+1%
ポイント+1%
商品説明





添付スプーン1杯あたりの量 | カロリー | たんぱく質量 | たんぱく質割合 | プロテインの種類・製法 |
約31g | 120kcal | 24g | 77.4% | ・WPI(ホエイプロテインアイソレート) ・WPC(ホエイプロテインコンセントレート) ・ホエイペプチド |